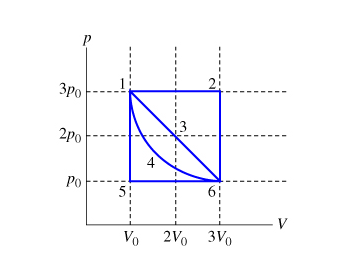
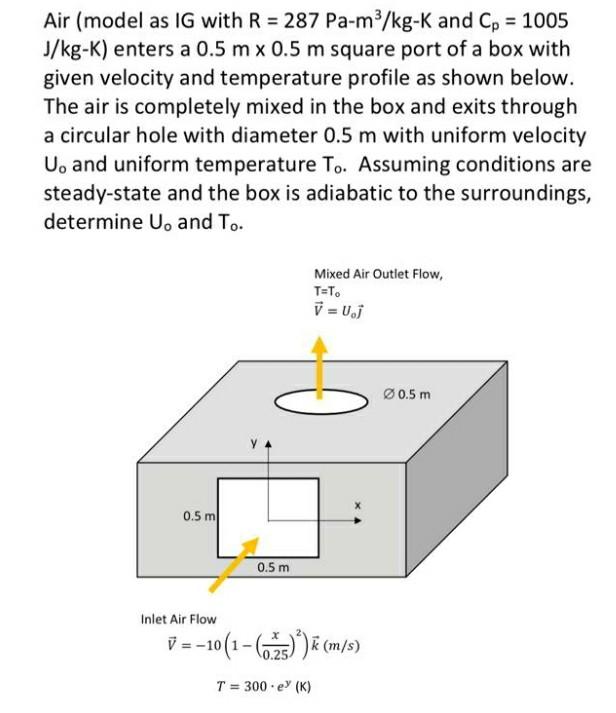
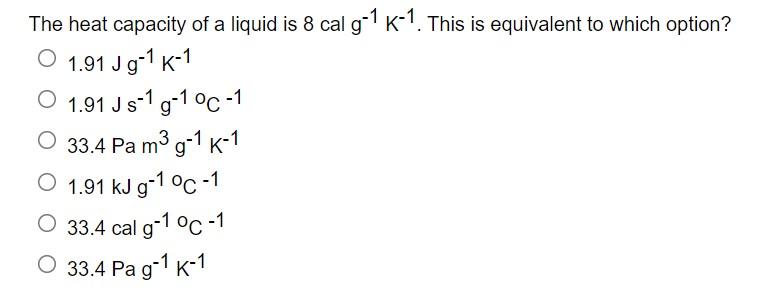Temperature and thermal expansion Specific Heat Capacity Phase changes and Heat Molecular picture of a gas Ideal gas law Kinetic theory of. - ppt download

Unit One Quiz Solutions and Unit Two Goals Mechanical Engineering 370 Thermodynamics Larry Caretto February 11, ppt download

Dose-dependent HASMC activation by Pam 3 CSK 4 , poly(I:C), LPS, R-837... | Download Scientific Diagram
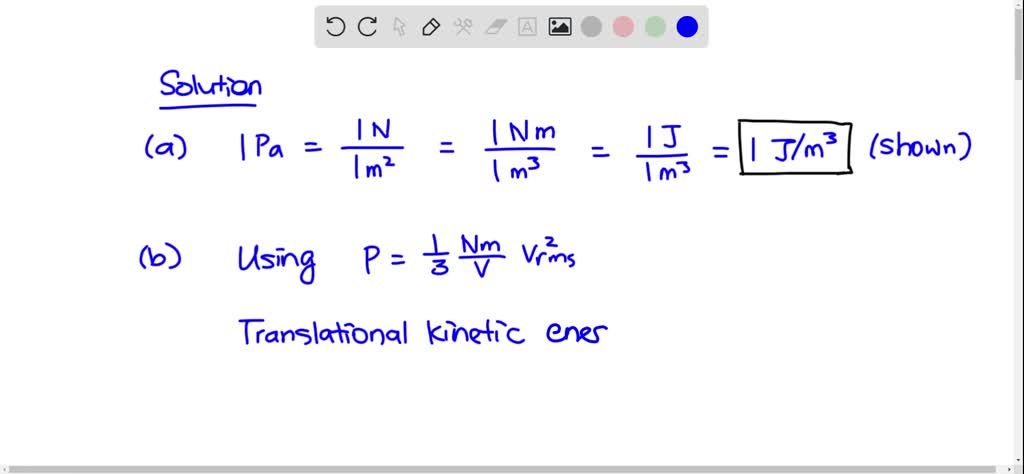
SOLVED:(a) Show that 1 Pa 1 J/m3. (b) Show that the density in space of the translational kinetic energy of an ideal gas is 3P/2.

SOLVED: A sample of an ideal gas is initially at a volume of 3.5 L. The gas expands to a volume of 7.0 m3 when 2 J of heat is applied to

SOLVED: Question A rigid tank of 0.2 m3 initially contains superheated steam at 300 'C and 3MPa is heated using an external source. A valve at the top of the tank is