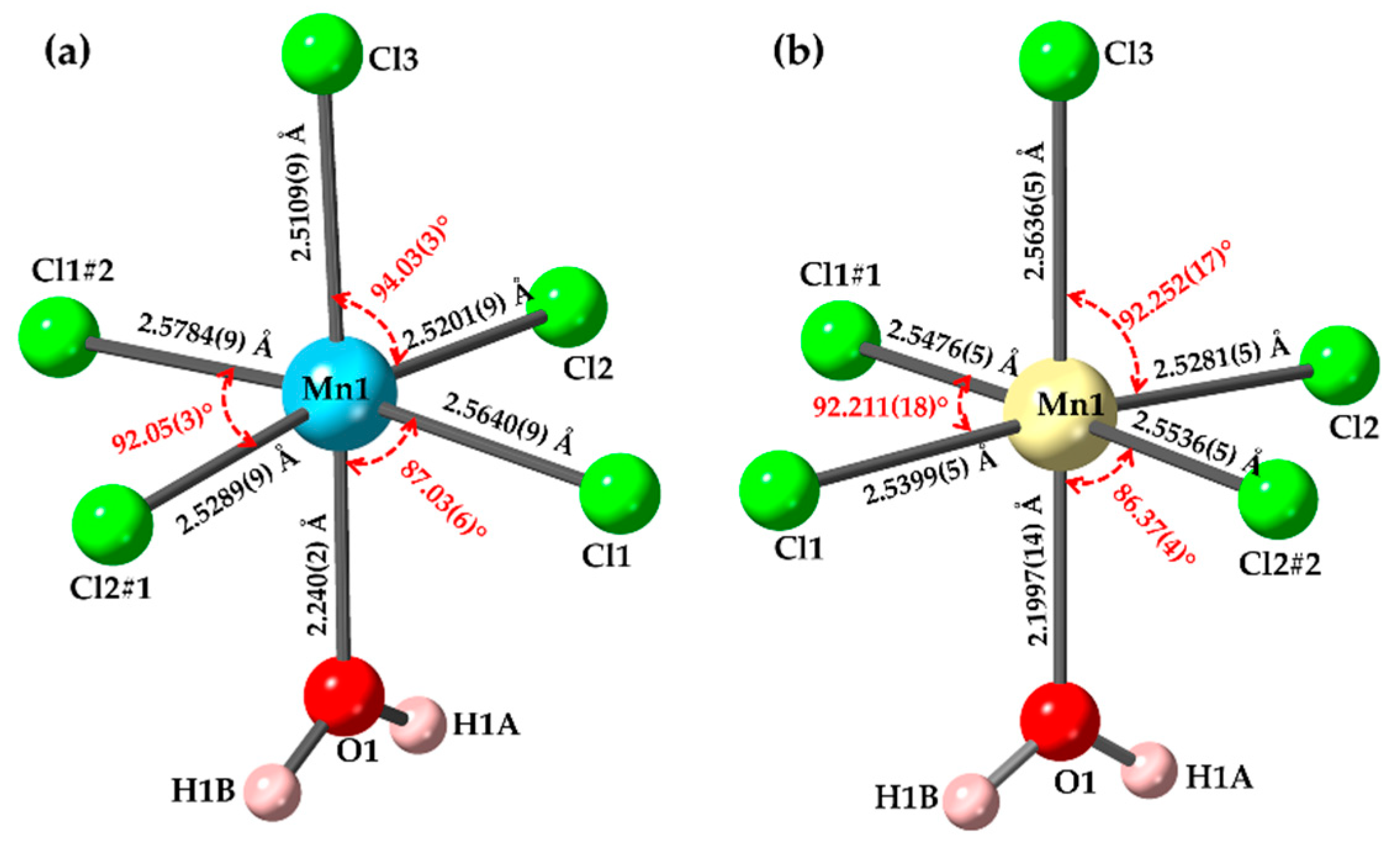
Crystals | Free Full-Text | Structural Variations in Manganese Halide Chain Compounds Mediated by Methylimidazolium Isomers
![In solid state PCl5 exist as an ionic solid [PCl4]^+ [PCl6]^- hybridisation state of cation and anion respectively : In solid state PCl5 exist as an ionic solid [PCl4]^+ [PCl6]^- hybridisation state of cation and anion respectively :](https://d1hj4to4g9ba46.cloudfront.net/questions/124193_77117_ans.png)
In solid state PCl5 exist as an ionic solid [PCl4]^+ [PCl6]^- hybridisation state of cation and anion respectively :

Devise the Lewis formula, sketch the three-dimensional shape, and name the electronic and ionic geometries for the given ion. PCl6 - | Homework.Study.com

A) DSC curves obtained for neat PCL, PCL3, and PCL6 during the second... | Download Scientific Diagram







![PCl5 is highly unstable and in solid state it exists as into [PCl4]+ and [ PCl6 PCl5 is highly unstable and in solid state it exists as into [PCl4]+ and [ PCl6](https://www.gkseries.com/blog/wp-content/uploads/2022/04/PCl5-is-highly-unstable-and-in-solid-state-it-exists-as-into-PCl4-and-PCl6-ions.-The-geometry-of-PCl6-is.png)
![What is the molecular structure of [PCl_6]^-, and what is the bond angle? | Homework.Study.com What is the molecular structure of [PCl_6]^-, and what is the bond angle? | Homework.Study.com](https://homework.study.com/cimages/multimages/16/pcl62958494712247179425.png)





