Alkylation of Phenol with Tertiary Butyl Alcohol over Zeolites | Organic Process Research & Development
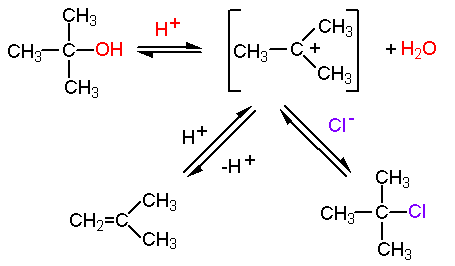
Assertion: Tert. butyl alcohol on heating with conc. H2SO4 at 413 K gives 2 methyl propane as the main product and not di tert. butyl ether. Reason: All alcohols are readily dehydrated
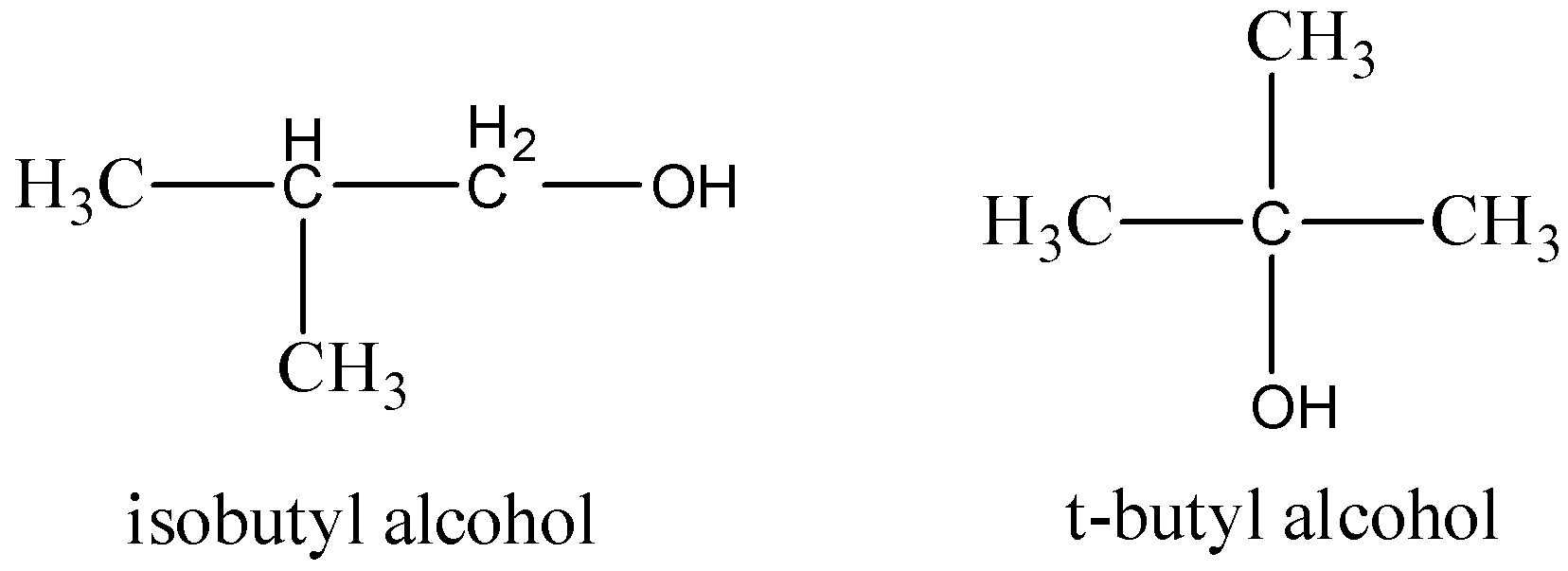
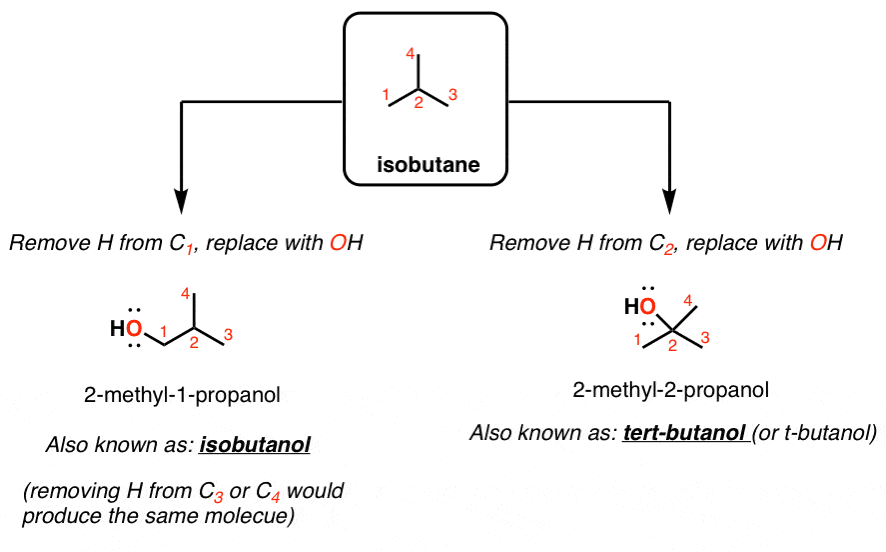
Which of the following pairs of compounds are position isomers?A.Isobutyl alcohol and s-butyl alcoholB.Isobutyl alcohol and t-butyl alcoholC.Isobutyl alcohol and neo-pentyl alcoholD.Ethyl alcohol and ethylene glycol

When the vapours of tertiary butyl alcohol are passed through heated copper at 573 K , the product formed is:
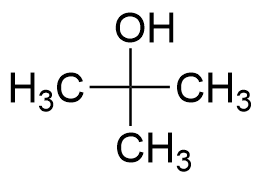
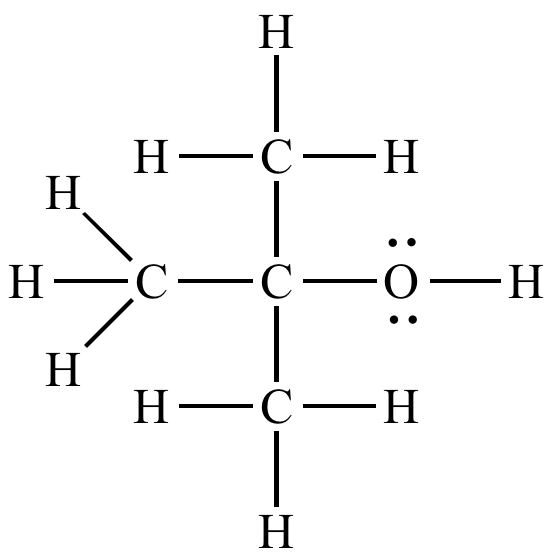
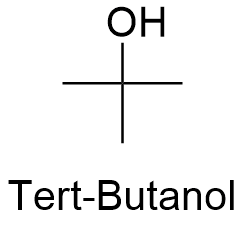
75-65-0 | Tert-butyl Alcohol | 1,1-Dimethylethanol; 2-Methyl-2-propanol; Trimethylcarbinol; Trimethylmethanol; t-Butanol; tert-Butanol | C₄H₁₀O | TRC

53001-22-2 | tert-Butyl Alcohol-d10 | 2-Methyl-2-propanol-d10; tert-Butanol-d10; 2-(Methyl-d3)-2-propan-1,1,1,3,3,3-d6-ol-d; Trimethylcarbinol-d10; Trimethylmethanol-d10; t-Butanol-d10; | C₄D₁₀O | TRC

t-butyl alcohol reacts less rapidly with metalic sodium than the primary alcohol. Explain why? - YouTube