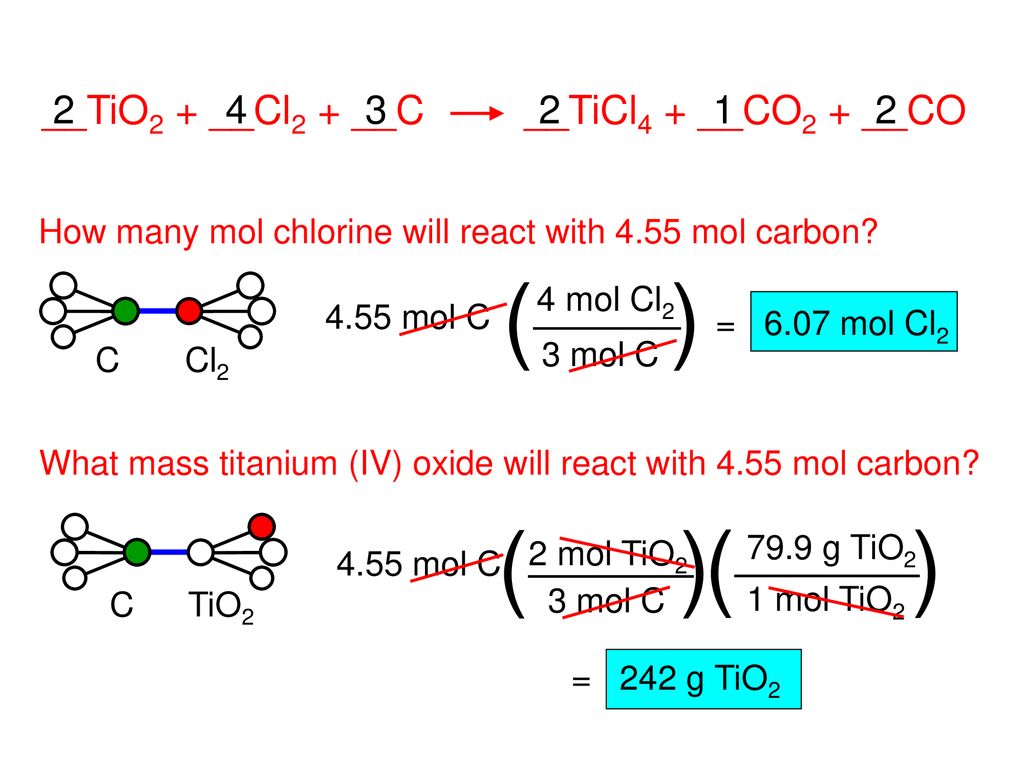
SOLVED: Challenge Titanium is a transition metal used in many alloys because it is extremely strong and lightweight. Titanium tetrachloride (TiCl4) is extracted from titanium oxide (TiO2) using chlorine and coke (carbon).

First-Principles Calculations of Adsorption Reactions of C and Cl2 on TiO2 (001) Surface with Bridge-Oxygen Defect in Fluidized Chlorination | SpringerLink
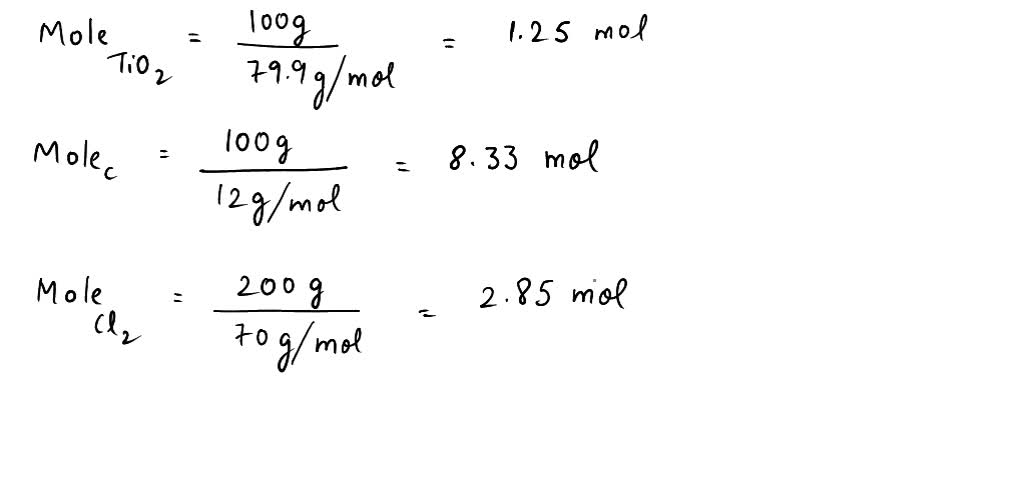
SOLVED: Titanium(IV) chloride can be be prepared from titanium(IV) oxide, carbon and chlorine gas according to the following chemical equation: 3TiO2(s) + 4 C(s) + 6 Cl2(g) ⟶ 3 TiCl4(l) + 2

SOLVED: (a) Write the balanced equation TiO2+C+2 Cl2→TiCl4+CO2 (b) Calculate the molar mass of TiO2 show your math below. (c) How many moles of Cl2 gas are needed to react with 1.25

Dependence of Cl 2 concentration as a function of (a) amount of coated... | Download Scientific Diagram

5.75 | The white pigment TiO2 is prepared by the reaction of titanium tetrachloride, TiCl4, with - YouTube

Insight into the performance of UV/chlorine/TiO2 on carbamazepine degradation: The crucial role of chlorine oxide radical (ClO•) - ScienceDirect
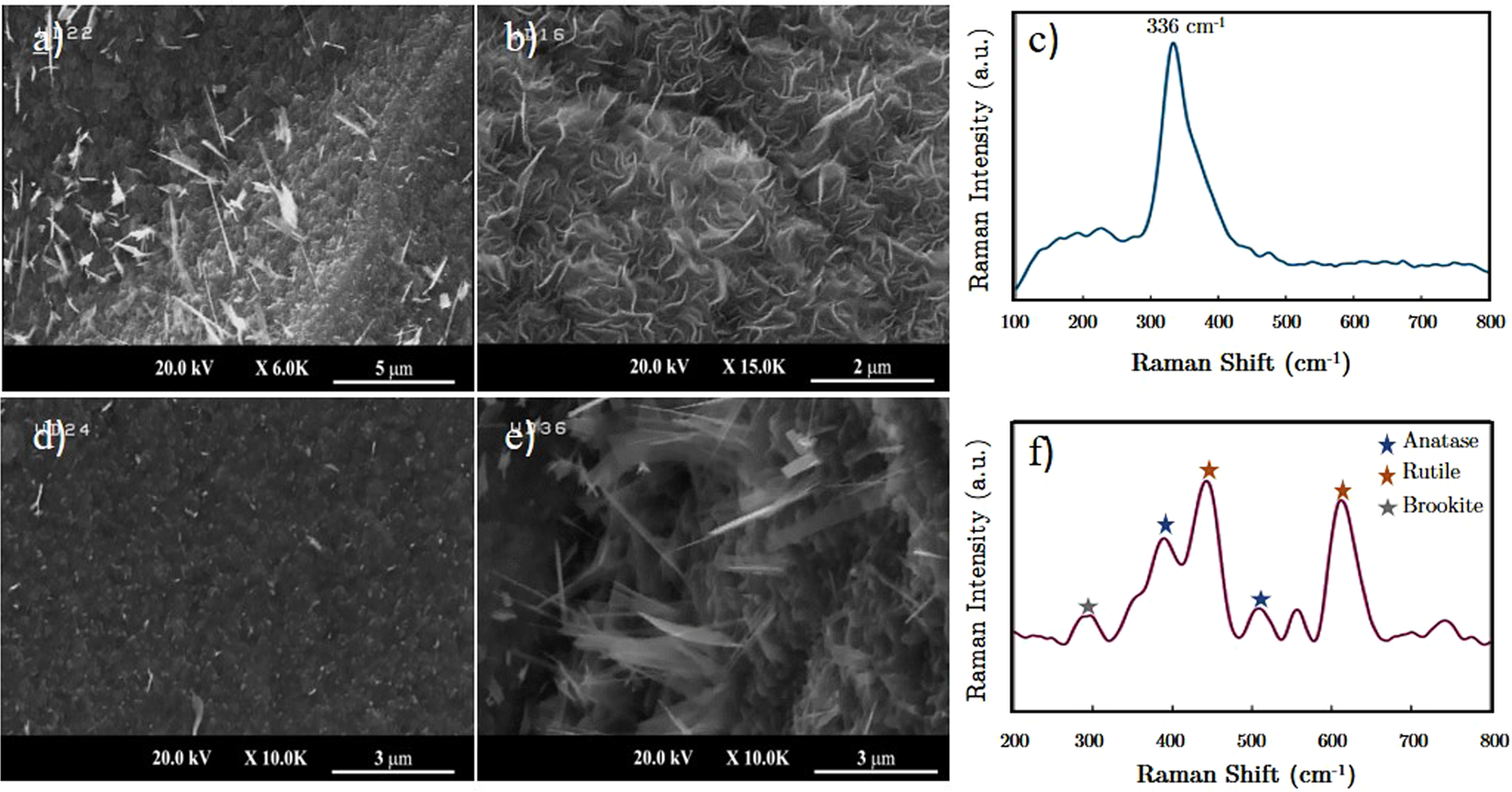
Evolution of large area TiS2-TiO2 heterostructures and S-doped TiO2 nano-sheets on titanium foils | Scientific Reports

Catalysts | Free Full-Text | Heterogeneous Photocatalytic Chlorination of Methylene Blue Using a Newly Synthesized TiO2-SiO2 Photocatalyst